Upsher-Smith Launches Fluvoxamine Maleate Tablets
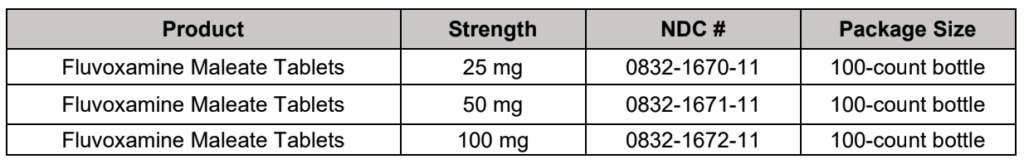
Maple Grove, MN – January 10, 2020 – Upsher-Smith Laboratories, LLC (Upsher-Smith) today announced the launch of Fluvoxamine Maleate Tablets in 25 mg, 50 mg and 100 mg strengths.
The fluvoxamine tablet market had U.S. sales of approximately $28 million for the 12 months ending September 2019 according to IQVIA.
Product Information
For questions about ordering, please call Upsher-Smith at 1-800-654-2299.
WARNING: SUICIDALITY AND ANTIDEPRESSANTS
See full prescribing information for complete boxed warning.
Increased risk of suicidal thinking and behavior in children, adolescents, and young adults taking antidepressants for major depressive disorder and other psychiatric disorders. Fluvoxamine maleate is not approved for use in pediatric patients except those with obsessive-compulsive disorder.
Please refer to the full Prescribing Information, including Boxed Warning for Fluvoxamine Maleate Tablets here. You can also call 1-888-650-3789 to obtain a copy of the full Prescribing Information.
You are encouraged to report suspected adverse reactions to Upsher-Smith Laboratories, LLC at 1-855-899-9180 or to the FDA by visiting http://www.fda.gov/medwatch.
About Upsher-Smith
Upsher-Smith Laboratories, LLC is a trusted U.S. pharmaceutical company that strives to improve the health and lives of patients through an unwavering commitment to high-quality products and sustainable growth. Since 1919, it has brought generics and brands to a wide array of customers backed by an attentive level of service, strong industry relationships, and dedication to uninterrupted supply.
As Upsher-Smith celebrates its 100-year anniversary, the company enters a new ambitious era that has been accelerated by its 2017 acquisition by Sawai Pharmaceutical Co., Ltd. Upsher-Smith and Sawai plan to leverage each other for growth worldwide and embark on an exciting new chapter as they seek to deliver the best value for their stakeholders, and most
importantly, Do More GoodTM for the patients they serve.
Do More Good is a trademark of Upsher-Smith Laboratories, LLC.
Contact: Elizabeth Likly
Kovak-Likly Communications
203-762-8833, elikly@klcpr.com

